Drug Catalog - Product Detail
ALENDRONATE SODIUM 35MG TAB 12CT
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 00115-1679-26 | AMNEAL PHARMACEUTICALS | 12 | 35MG | TABLET |
PACKAGE FILES





Generic Name
ALENDRONATE SODIUM
Substance Name
ALENDRONATE SODIUM
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA075710
Description
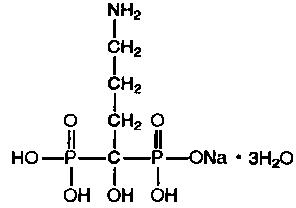
11 DESCRIPTION Alendronate Sodium Tablets, USP is a bisphosphonate that acts as a specific inhibitor of osteoclast-mediated bone resorption. Bisphosphonates are synthetic analogs of pyrophosphate that bind to the hydroxyapatite found in bone. Alendronate sodium, USP is chemically described as (4-amino-1-hydroxybutylidene) bisphosphonic acid monosodium salt trihydrate. The structural formula is: C 4 H 12 NNaO 7 P 2 •3H 2 O M.W. 325.12 Alendronate sodium, USP is a white, crystalline, nonhygroscopic powder. It is soluble in water, very slightly soluble in alcohol, and practically insoluble in chloroform. Each tablet for oral administration contains 6.53, 13.05, 45.68, 52.21 or 91.36 mg of alendronate monosodium salt trihydrate, which is the molar equivalent of 5, 10, 35, 40 and 70 mg, respectively, of free acid, and the following inactive ingredients: croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. Alendronate Sodium Tablets, USP meet USP Dissolution Test 2. image of alendronate sodium chemical structure
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING Alendronate Sodium Tablets USP, for oral administration, are available as: 5 mg – white to off-white, round, flat-faced beveled-edge, unscored tablet debossed with “93” on one side and “5140” on the other side. They are available as follows: Bottles of 30: NDC 0115-1676-08 Bottles of 100: NDC 0115-1676-01 10 mg – white to off-white, round, convex, unscored tablet debossed with “93” on one side and “5141” on the other side. They are available as follows: Bottles of 30: NDC 0115-1678-08 Bottles of 100: NDC 0115-1678-01 35 mg – white to off-white, pillow-shaped, convex, unscored tablet debossed with “93” on one side and “5172” on the other side. They are available as follows: Cartons of 4: NDC 0115-1679-34 Cartons of 12 (3 x 4): NDC 0115-1679-26 40 mg – white to off-white, oval, convex, unscored tablet debossed with “93” on one side and “5142” on the other side. They are available as follows: Bottles of 30: NDC 0115-1680-08 70 mg – white to off-white, pillow-shaped, convex, unscored tablet debossed with “93” on one side and “5171” on the other side. They are available as follows: Cartons of 4: NDC 0115-1681-34 Cartons of 12 (3 x 4): NDC 0115-1681-26 Storage Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required). KEEP THIS AND ALL OTHER MEDICATIONS OUT OF THE REACH OF CHILDREN.
Indications & Usage
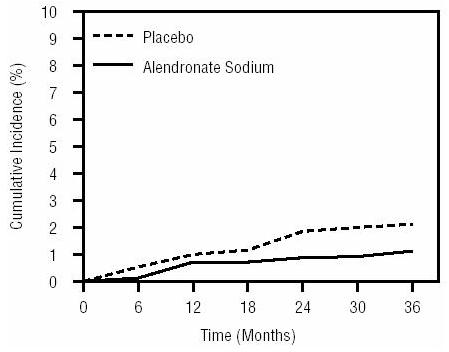
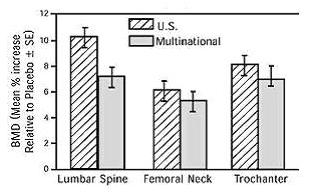
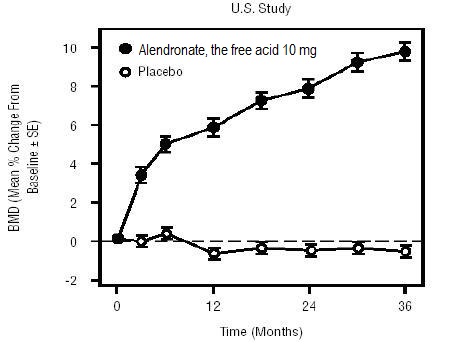
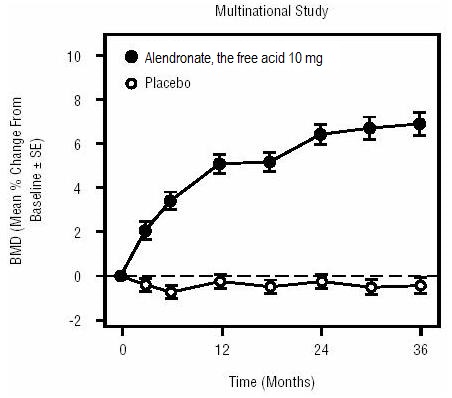
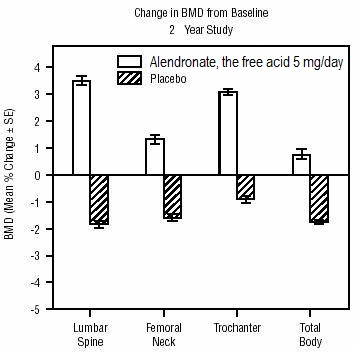
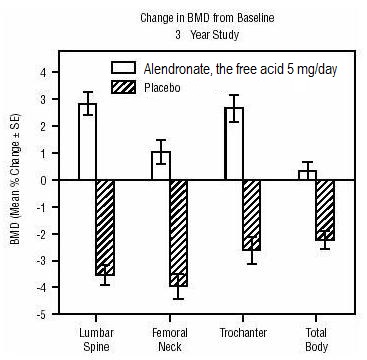
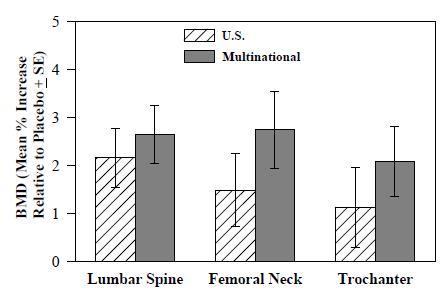
1 INDICATIONS AND USAGE Alendronate sodium tablets are a bisphosphonate indicated for: Treatment and prevention of osteoporosis in postmenopausal women ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone ( 1.5 ) Limitations of use: Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use. ( 1.6 ) 1.1 Treatment of Osteoporosis in Postmenopausal Women Alendronate sodium tablets are indicated for the treatment of osteoporosis in postmenopausal women. In postmenopausal women, alendronate sodium tablets increase bone mass and reduces the incidence of fractures, including those of the hip and spine (vertebral compression fractures) [s ee Clinical Studies (14.1) ]. 1.2 Prevention of Osteoporosis in Postmenopausal Women Alendronate sodium tablets are indicated for the prevention of postmenopausal osteoporosis [ see Clinical Studies (14.2) ]. 1.3 Treatment to Increase Bone Mass in Men with Osteoporosis Alendronate sodium tablets are indicated for treatment to increase bone mass in men with osteoporosis [ see Clinical Studies (14.3) ]. 1.4 Treatment of Glucocorticoid-Induced Osteoporosis Alendronate sodium tablets are indicated for the treatment of glucocorticoid-induced osteoporosis in men and women receiving glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and who have low bone mineral density [ see Clinical Studies (14.4) ]. 1.5 Treatment of Paget's Disease of Bone Alendronate sodium tablets are indicated for the treatment of Paget's disease of bone in men and women. Treatment is indicated in patients with Paget's disease of bone who have alkaline phosphatase at least two times the upper limit of normal, or those who are symptomatic, or those at risk for future complications from their disease [ see Clinical Studies (14.5) ]. 1.6 Important Limitations of Use The optimal duration of use has not been determined. The safety and effectiveness of alendronate sodium tablets for the treatment of osteoporosis are based on clinical data of four years duration. All patients on bisphosphonate therapy should have the need for continued therapy re-evaluated on a periodic basis. Patients at low-risk for fracture should be considered for drug discontinuation after 3 to 5 years of use. Patients who discontinue therapy should have their risk for fracture re-evaluated periodically.
Dosage and Administration
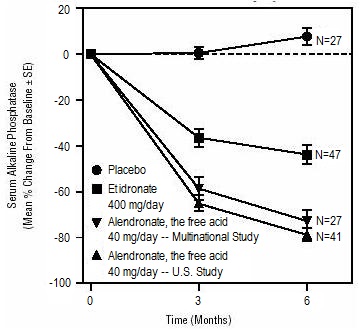
2 DOSAGE AND ADMINISTRATION Treatment of osteoporosis in postmenopausal women and in men: 10 mg daily or 70 mg (tablet) once weekly. ( 2.1 , 2.3 ) Prevention of osteoporosis in postmenopausal women: 5 mg daily or 35 mg once weekly. ( 2.2 ) Glucocorticoid-induced osteoporosis: 5 mg daily; or 10 mg daily in postmenopausal women not receiving estrogen. ( 2.4 ) Paget's disease: 40 mg daily for six months. ( 2.5 ) Instruct patients to: ( 2.6 ) Swallow tablets whole with 6 to 8 ounces plain water at least 30 minutes before the first food, drink, or medication of the day. Not lie down for at least 30 minutes after taking alendronate sodium tablets and until after food. 2.1 Treatment of Osteoporosis in Postmenopausal Women The recommended dosage is: one 70 mg tablet once weekly or one 10 mg tablet once daily 2.2 Prevention of Osteoporosis in Postmenopausal Women The recommended dosage is: one 35 mg tablet once weekly or one 5 mg tablet once daily 2.3 Treatment to Increase Bone Mass in Men with Osteoporosis The recommended dosage is: one 70 mg tablet once weekly or one 10 mg tablet once daily 2.4 Treatment of Glucocorticoid-Induced Osteoporosis The recommended dosage is one 5 mg tablet once daily, except for postmenopausal women not receiving estrogen, for whom the recommended dosage is one 10 mg tablet once daily. 2.5 Treatment of Paget's Disease of Bone The recommended treatment regimen is 40 mg once a day for six months. Re-treatment of Paget's Disease Re-treatment with alendronate sodium tablets may be considered, following a six-month post-treatment evaluation period in patients who have relapsed, based on increases in serum alkaline phosphatase, which should be measured periodically. Re-treatment may also be considered in those who failed to normalize their serum alkaline phosphatase. 2.6 Important Administration Instructions Instruct patients to do the following: Take alendronate sodium tablets at least one-half hour before the first food, beverage, or medication of the day with plain water only [ see Patient Counseling Information (17.2) ] . Other beverages (including mineral water), food, and some medications are likely to reduce the absorption of alendronate sodium tablets [ see Drug Interactions (7.1) ] . Waiting less than 30 minutes, or taking alendronate sodium tablets with food, beverages (other than plain water) or other medications will lessen the effect of alendronate sodium tablets by decreasing its absorption into the body. Take alendronate sodium tablets upon arising for the day. To facilitate delivery to the stomach and thus reduce the potential for esophageal irritation, a alendronate sodium tablet should be swallowed with a full glass of water (6 to 8 ounces). Patients should not lie down for at least 30 minutes and until after their first food of the day. Alendronate sodium tablets should not be taken at bedtime or before arising for the day. Failure to follow these instructions may increase the risk of esophageal adverse experiences [ see Warnings and Precautions (5.1) and Patient Counseling Information (17.2) ]. 2.7 Recommendations for Calcium and Vitamin D Supplementation Instruct patients to take supplemental calcium if dietary intake is inadequate [ see Warnings and Precautions (5.2) ]. Patients at increased risk for vitamin D insufficiency (e.g., over the age of 70 years, nursing home-bound, or chronically ill) may need vitamin D supplementation. Patients with gastrointestinal malabsorption syndromes may require higher doses of vitamin D supplementation and measurement of 25-hydroxyvitamin D should be considered. Patients treated with glucocorticoids should receive adequate amounts of calcium and vitamin D. 2.8 Administration Instructions for Missed Doses If a once-weekly dose of alendronate sodium tablets is missed, instruct patients to take one dose on the morning after they remember. They should not take two doses on the same day but should return to taking one dose once a week, as originally scheduled on their chosen day .
