Drug Catalog - Product Detail
BETHANECHOL CHLORIDE Tab 50 MG 100EA
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 65162-0574-10 | AMNEAL PHARMACEUTICALS | 100 | 50MG | TABLET |
PACKAGE FILES
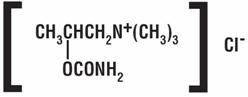




Generic Name
BETHANECHOL CHLORIDE
Substance Name
BETHANECHOL CHLORIDE
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA040855
Description
DESCRIPTION Bethanechol chloride, a cholinergic agent, is a synthetic ester which is structurally and pharmacologically related to acetylcholine. It is designated chemically as 2-[(aminocarbonyl) oxy]- N, N, N -trimethyl-1-propanaminium chloride. Its molecular formula is C 7 H 17 CIN 2 0 2 and its structural formula is: It is a white, hygroscopic crystalline powder having a slight amine-like odor, freely soluble in water, and has a molecular weight of 196.68. Each tablet for oral administration contains 5 mg, 10 mg, 25 mg or 50 mg bethanechol chloride, USP. Tablets also contain the following inactive ingredients: anhydrous lactose, colloidal silicon dioxide, magnesium stearate, microcrystalline cellulose and sodium starch glycolate. The 25 mg and 50 mg tablets also contain D&C Yellow #10 Aluminum Lake and FD&C Yellow #6 Aluminum Lake. 38055a93-figure-01
How Supplied
HOW SUPPLIED Bethanechol Chloride Tablets, USP, 5 mg are supplied as white, round, flat-faced, bevelled tablets debossed with ‘AN’ above bisect and ‘571’ below bisect on one side and plain on the other side. They are available as follows: Bottle of 100: NDC 65162-571-10 Bottle of 500: NDC 65162-571-50 Bottle of 1000: NDC 65162-571-11 Bethanechol Chloride Tablets, USP, 10 mg are supplied as white, round, flat-faced, bevelled tablets debossed with ‘AN’ above bisect and ‘572’ below bisect on one side and plain on the other side. They are available as follows: Bottle of 100: NDC 65162-572-10 Bottle of 500: NDC 65162-572-50 Bottle of 1000: NDC 65162-572-11 Bethanechol Chloride Tablets, USP, 25 mg are supplied as yellow, round, flat-faced, bevelled tablets debossed with ‘AN’ above bisect and ‘573’ below bisect on one side and plain on the other side. They are available as follows: Bottle of 100: NDC 65162-573-10 Bottle of 500: NDC 65162-573-50 Bottle of 1000: NDC 65162-573-11 Bethanechol Chloride Tablets, USP, 50 mg are supplied as yellow, round, flat-faced, bevelled tablets debossed with ‘AN’ above bisect and ‘574’ below bisect on one side and plain on the other side. They are available as follows: Bottle of 100: NDC 65162-574-10 Bottle of 500: NDC 65162-574-50 Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. This container is not intended for household use. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required). KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN. Distributed by: Amneal Pharmaceuticals LLC Bridgewater, NJ 08807 Rev. 05-2018-03
Indications & Usage
INDICATIONS AND USAGE Bethanechol chloride tablets are indicated for the treatment of acute postoperative and postpartum nonobstructive (functional) urinary retention and for neurogenic atony of the urinary bladder with retention.
Dosage and Administration
DOSAGE AND ADMINISTRATION Dosage must be individualized, depending on the type and severity of the condition to be treated. Preferably give the drug when the stomach is empty. If taken soon after eating, nausea and vomiting may occur. The usual adult oral dose ranges from 10 to 50 mg three or four times a day. The minimum effective dose is determined by giving 5 to 10 mg initially, and repeating the same amount at hourly intervals until satisfactory response occurs, or until a maximum of 50 mg has been given. The effects of the drug sometimes appear within 30 minutes, and are usually maximal within 60 to 90 minutes. The drug effects persist for about one hour. If necessary, the effects of the drug can be abolished promptly by atropine (see OVERDOSAGE ).
