Drug Catalog - Product Detail
CANDESARTAN CILEXETIL HCTZ TB 32/12.5MG 90
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 00378-3002-77 | MYLAN | 90 | 32-12.5MG | TABLET |
PACKAGE FILES



Generic Name
CANDESARTAN CILEXETIL AND HYDROCHLOROTHIAZIDE
Substance Name
CANDESARTAN CILEXETIL
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA090704
Description
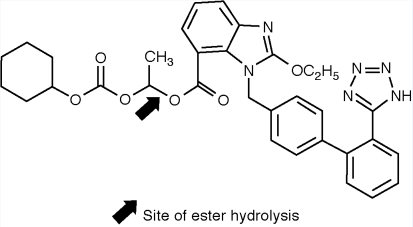
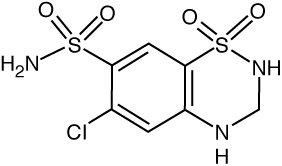
DESCRIPTION Candesartan cilexetil and hydrochlorothiazide tablets, USP combine an angiotensin II receptor (type AT 1 ) antagonist and a diuretic, hydrochlorothiazide. Candesartan cilexetil, a nonpeptide, is chemically described as (±)2-Ethoxy-1-[[2’-(1 H -tetrazol-5-yl)[1,1’-biphenyl]-4-yl]methyl]-1 H -benzimidazole-7-carboxylic acid-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl ester. Its molecular formula is C 33 H 34 N 6 O 6 , and its structural formula is: Candesartan cilexetil, USP is a white to off-white powder with a molecular weight of 610.66. It is practically insoluble in water and sparingly soluble in methanol. Candesartan cilexetil is a racemic mixture containing one chiral center at the cyclohexyloxycarbonyloxy ethyl ester group. Following oral administration, candesartan cilexetil undergoes hydrolysis at the ester link to form the active drug, candesartan, which is achiral. Hydrochlorothiazide is 2H-1,2,4-Benzothiadiazine-7-sulfonamide, 6-chloro-3,4-dihydro-,1,1-dioxide. Its molecular formula is C 7 H 8 ClN 3 O 4 S 2 and its structural formula is: Hydrochlorothiazide, USP is a white, or practically white, practically odorless crystalline powder with a molecular weight of 297.7, which is slightly soluble in water, but freely soluble in sodium hydroxide solution. Candesartan cilexetil and hydrochlorothiazide tablets are available for oral administration in three tablet strengths of candesartan cilexetil and hydrochlorothiazide. Candesartan cilexetil and hydrochlorothiazide tablets contain 16 mg or 32 mg of candesartan cilexetil and 12.5 mg or 25 mg of hydrochlorothiazide providing for the following available combinations: 16 mg/12.5 mg, 32 mg/12.5 mg or 32 mg/25 mg. The inactive ingredients of the tablets are carboxymethylcellulose calcium, corn starch, glyceryl stearate, hydroxypropyl cellulose, lactose monohydrate, magnesium stearate and yellow iron oxide. The 16 mg/12.5 mg tablet and 32 mg/25 mg tablet also contain red iron oxide as a colorant. Candesartan Cilexetil Stuctural Formula Hydrochlorothiazide Structural Formula
How Supplied
HOW SUPPLIED Candesartan Cilexetil and Hydrochlorothiazide Tablets, USP are available containing 16 mg or 32 mg of candesartan cilexetil, USP and 12.5 mg or 25 mg of hydrochlorothiazide, USP providing for the following available combinations: 16 mg/12.5 mg, 32 mg/12.5 mg or 32 mg/25 mg. The 16 mg/12.5 mg tablets are peach, mottled, round, scored tablets debossed with M on the left side of the score and X on the right side of the score on one side of the tablet and C1 on the other side of the tablet. They are available as follows: NDC 0378-3001-77 bottles of 90 tablets NDC 0378-3001-05 bottles of 500 tablets The 32 mg/12.5 mg tablets are yellow, mottled, round, scored tablets debossed with M on the left side of the score and X on the right side of the score on one side of the tablet and C2 on the other side of the tablet. They are available as follows: NDC 0378-3002-77 bottles of 90 tablets NDC 0378-3002-05 bottles of 500 tablets The 32 mg/25 mg tablets are peach, mottled, round, scored tablets debossed with M on the left of the score and X on the right of the score on one side of the tablet and C3 on the other side of the tablet. They are available as follows: NDC 0378-3003-77 bottles of 90 tablets NDC 0378-3003-05 bottles of 500 tablets Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. Keep container tightly closed. Manufactured for: Mylan Pharmaceuticals Inc. Morgantown, WV 26505 U.S.A. Manufactured by: Mylan Laboratories Limited Hyderabad — 500 096, India 75076992 Revised: 8/2020 MX:CNDHTZ:R10
Indications & Usage
INDICATIONS AND USAGE Candesartan cilexetil and hydrochlorothiazide tablets are indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including the class to which this drug principally belongs. There are no controlled trials demonstrating risk reduction with candesartan cilexetil and hydrochlorothiazide tablets. Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC). Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly. Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal. Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy. This fixed dose combination is not indicated for initial therapy (see DOSAGE AND ADMINISTRATION ).
Dosage and Administration
DOSAGE AND ADMINISTRATION The usual recommended starting dose of candesartan cilexetil is 16 mg once daily when it is used as monotherapy in patients who are not volume depleted. Candesartan cilexetil tablets can be administered once or twice daily with total daily doses ranging from 8 mg to 32 mg. Patients requiring further reduction in blood pressure should be titrated to 32 mg. Doses larger than 32 mg do not appear to have a greater blood pressure lowering effect. Hydrochlorothiazide is effective in doses of 12.5 to 50 mg once daily. Use in Renal Impairment Dosing recommendations for candesartan cilexetil and hydrochlorothiazide tablets in patients with creatinine clearance < 30 mg/min cannot be provided (see SPECIAL POPULATIONS: Renal Insufficiency ). Use in Moderate to Severe Hepatic Impairment Candesartan cilexetil and hydrochlorothiazide tablets are not recommended for initiation because the appropriate starting dose, 8 mg, cannot be given (see SPECIAL POPULATIONS, Hepatic Insufficiency ). Replacement Therapy The combination may be substituted for the titrated components. Dose Titration by Clinical Effect A patient whose blood pressure is not controlled on 25 mg of hydrochlorothiazide once daily can expect an incremental effect from candesartan cilexetil and hydrochlorothiazide tablets 16 mg/12.5 mg. A patient whose blood pressure is controlled on 25 mg of hydrochlorothiazide but is experiencing decreases in serum potassium can expect the same or incremental blood pressure effects from candesartan cilexetil and hydrochlorothiazide tablets 16 mg/12.5 mg and serum potassium may improve. A patient whose blood pressure is not controlled on 32 mg of candesartan cilexetil can expect incremental blood pressure effects from candesartan cilexetil and hydrochlorothiazide tablets 32 mg/12.5 mg and then 32 mg/25 mg. The maximal antihypertensive effect of any dose of candesartan cilexetil and hydrochlorothiazide tablets can be expected within 4 weeks of initiating that dose. Candesartan cilexetil and hydrochlorothiazide tablets may be administered with other antihypertensive agents. Candesartan cilexetil and hydrochlorothiazide tablets may be administered with or without food.
