Drug Catalog - Product Detail
CEFAZOLIN SODIUM FOR INJECTION INJECT. 1GM/ML 25X10ML
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 00143-9924-90 | HIKMA | 1 | 1GM | SOLUTION |
PACKAGE FILES


Generic Name
CEFAZOLIN
Substance Name
CEFAZOLIN SODIUM
Product Type
HUMAN PRESCRIPTION DRUG
Route
INTRAVENOUS
Application Number
ANDA065143
Description
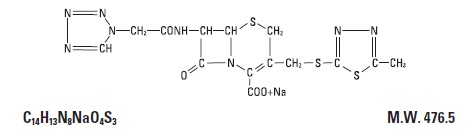
DESCRIPTION Cefazolin for Injection, USP is a semi-synthetic cephalosporin for parenteral administration. It is the sodium salt of (6R, 7R)-3-[[(5-methyl-1,3,4-thiadiazol-2-yl)]thio]methyl]-8-oxo-7-[2(1H-tetrazol-1-yl)acetoamido]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid. The structural formula is as follows: Cefazolin for Injection, USP is a white to cream sterile powder. The color of Cefazolin for Injection, USP solutions may range from pale yellow to yellow without a change in potency. The pH ranges from 4.0 and 6.0 for a solution containing 100 mg of cefazolin per mL. Cefazolin for Injection, USP is supplied in 10 or 20 grams Pharmacy Bulk Packages. Each Pharmacy Bulk Package contains Cefazolin Sodium, USP equivalent to 10 or 20 grams of cefazolin. The sodium content is approximately 48 mg (2.1 mEq) per gram of cefazolin sodium. It is to be administered by intravenous route. A Pharmacy Bulk Package is a container of a sterile preparation for intravenous use that contains many single doses. The contents are intended for use in a pharmacy admixture service and are restricted to the preparation of admixtures for intravenous infusion. FURTHER DILUTION IS REQUIRED BEFORE USE . Cef Chemical Structure
How Supplied
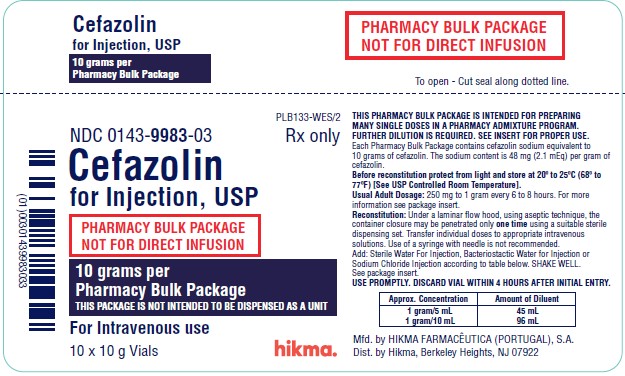
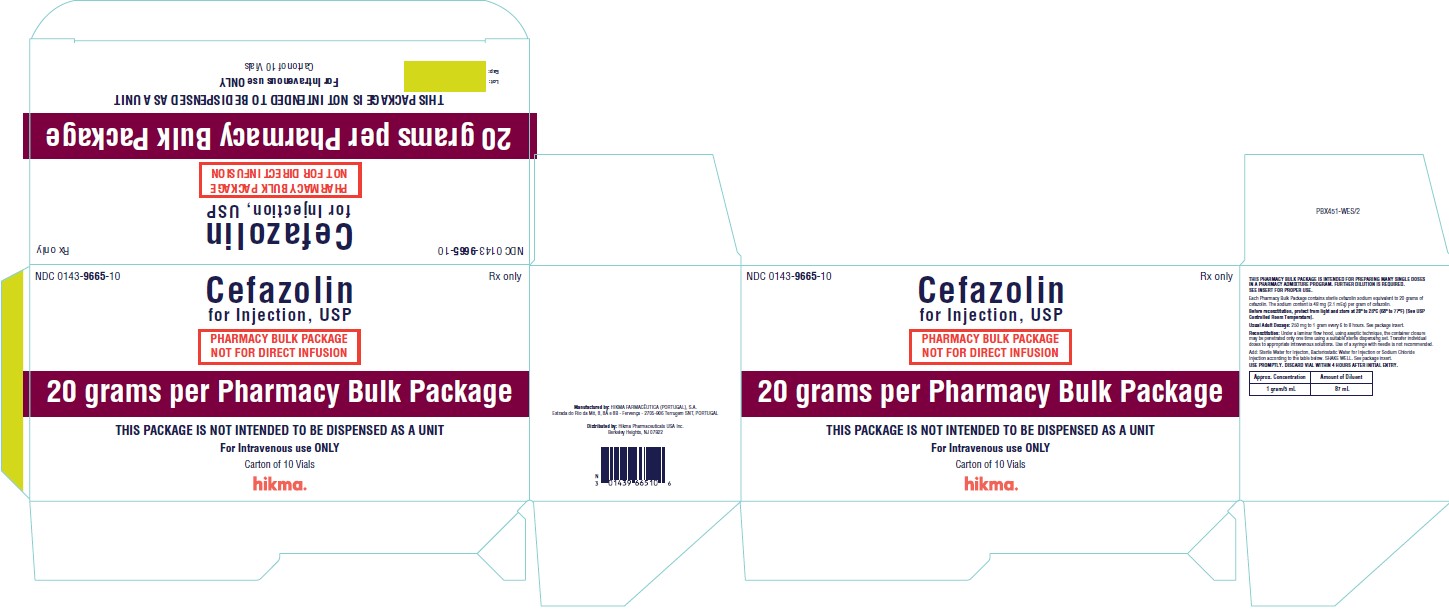
HOW SUPPLIED Cefazolin for Injection, USP, is supplied in 10 and 20 grams Pharmacy Bulk Package vials. Each Pharmacy Bulk Package contains cefazolin sodium equivalent to 10 or 20 grams of cefazolin, as follows: Cefazolin for Injection, USP Vial Size Packaged NDC No. 10 grams 100 mL Carton of 10 vials 0143-9983-03 20 grams 100 mL Carton of 10 vials 0143-9665-10 Also available as: Cefazolin for Injection, USP Vial Size Packaged NDC No. 500 mg 10 mL Carton of 25 vials 0143-9923-90 1 gram 10 mL Carton of 25 vials 0143-9924-90 Preservative Free. As with other cephalosporins, Cefazolin for Injection, USP tends to darken depending on storage conditions; within the stated recommendations; however, product potency is not adversely affected. Before reconstitution, protect from light and store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature]. This container closure is not made with natural rubber latex. CLINITEST is a registered trademark of Miles, Inc. CLINISTIX is a registered trademark of Bayer Corporation. Manufactured by: HIKMA FARMACÊUTICA (PORTUGAL) S.A. Estrada do Rio da Mó, nº8, 8A and 8B – Fervença 2705-906 Terrugem SNT PORTUGAL Distributed by: Hikma Pharmaceuticals USA Inc. Berkeley Heights, NJ 07922 Revised: September 2020 PIN089-WES/7
Indications & Usage
INDICATIONS AND USAGE Cefazolin for Injection, USP is indicated in the treatment of the following serious infections due to susceptible organisms: Respiratory Tract Infections: due to S. penumoniae, Klebsiella species, H. Influenzae, S. aureus (penicillin-sensitive and penicillin-resistant) and group A beta-hemolytic streptococci . Injectable benzathine penicillin is considered to be the drug of choice in treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever. Cefazolin for Injection, USP is effective in the eradication of streptococci from the nasopharynx; however, data establishing the efficacy of Cefazolin for Injection, USP in the subsequent prevention of rheumatic fever are not available. Urinary Tract Infections: due to E. coli, P. mirabilis, Klebsiella species, and some strains of enterobacter and enterococci . Skin and Skin Structure Infections: due to S. aureus (penicillin-sensitive and penicillin-resistant), group A beta-hemolytic streptococci, and other strains of streptococci. Biliary Tract Infections: due to E. coli , various strains of streptococci, P. mirabilis, Klebsiella species and S. aureus . Bone and Joint Infections: due to S. aureus . Genital Infections: (i.e., prostatitis, epididymitis) due to E. coli, P. mirabilis, Klebsiella species, and some strains of enterococci . Septicemia: due to S. pneumoniae , S. aureus (penicillin-sensitive and penicillin-resistant), P. mirabilis , E. coli, and Klebsiella species. Endocarditis: due to S. aureus (penicillin-sensitive and penicillin-resistant) and group A beta-hemolytic streptococci . Perioperative Prophylaxis: The prophylactic administration of Cefazolin for Injection, USP preoperatively, intraoperatively and postoperatively may reduce the incidence of certain postoperative infections in patients undergoing surgical procedures which are classified as contaminated or potentially contaminated (e.g., vaginal hysterectomy, and cholecystectomy in high‑risk patients such as those older than 70 years, with acute cholecystitis, obstructive jaundice or common duct bile stones). The perioperative use of Cefazolin for Injection, USP may also be effective in surgical patients in whom infection at the operative site would present a serious risk (e.g., during open-heart surgery and prosthetic arthroplasty). The prophylactic administration of Cefazolin for Injection, USP should usually be discontinued within a 24 hour period after the surgical procedure. In surgery where the occurrence of infection may be particularly devastating (e.g., open-heart surgery and prosthetic arthroplasty), the prophylactic administration of Cefazolin for Injection, USP may be continued for 3 to 5 days following the completion of surgery. If there are signs of infection, specimens for cultures should be obtained for the identification of the causative organism so that appropriate therapy may be instituted (see DOSAGE AND ADMINISTRATION ). To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefazolin for Injection, USP and other antibacterial drugs, Cefazolin for Injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Dosage and Administration
DOSAGE AND ADMINISTRATION Usual Adult Dosage Type of Infection Dose Frequency Moderate to severe infections 500 mg to 1 gram every 6 to 8 hours Mild infections caused by susceptible gram-positive cocci 250 mg to 500 mg every 8 hours Acute, uncomplicated urinary tract infections 1 gram every 12 hours Pneumococcal pneumonia 500 mg every 12 hours Severe, life-threatening infections (e.g., endocarditis, septicemia) In rare instances, doses of up to 12 grams of Cefazolin for Injection per day have been used. 1 gram to 1.5 grams every 6 hours Perioperative Prophylactic Use To prevent postoperative infection in contaminated or potentially contaminated surgery, recommended doses are: 1 gram IV administered ½ hour to 1 hour prior to the start of surgery. For lengthy operative procedures (e.g., 2 hours or more), 500 mg to 1 gram IV during surgery (administration modified depending on the duration of the operative procedure). 500 mg to 1 gram IV every 6 to 8 hours for 24 hours postoperatively. It is important that (1) the preoperative dose be given just (1/2 to 1 hour) prior to the start of surgery so that adequate antibiotic levels are present in the serum and tissues at the time of initial surgical incision; and (2) Cefazolin for Injection be administered, if necessary, at appropriate intervals during surgery to provide sufficient levels of the antibiotic at the anticipated moments of greatest exposure to infective organisms. In surgery where the occurrence of infection may be particularly devastating (e.g., open-heart surgery and prosthetic arthroplasty), the prophylactic administration of Cefazolin for Injection may be continued for 3 to 5 days following the completion of surgery. Dosage Adjustment for Patients with Reduced Renal Function Cefazolin for injection may be used in patients with reduced renal function with the following dosage adjustments: Patients with a creatinine clearance of 55 mL/min or greater or a serum creatinine of 1.5 mg % or less can be given full doses. Patients with creatinine clearance rates of 35 to 54 mL/min or serum creatinine of 1.6 to 3 mg % can also be given full doses but dosage should be restricted to at least 8 hour intervals. Patients with creatinine clearance rates of 11 to 34 mL/min or serum creatinine of 3.1 to 4.5 mg % should be given 1/2 the usual dose every 12 hours. Patients with creatinine clearance rates of 10 mL/min or less or serum creatinine of 4.6 mg % or greater should be given 1/2 the usual dose every 18 to 24 hours. All reduced dosage recommendations apply after an initial loading dose appropriate to the severity of the infection. Patients undergoing peritoneal dialysis: See CLINICAL PHARMACOLOGY . Pediatric Dosage In pediatric patients, a total daily dosage of 25 to 50 mg/kg (approximately 10 to 20 mg/lb) of body weight, divided into 3 or 4 equal doses, is effective for most mild to moderately severe infections. Total daily dosage may be increased to 100 mg/kg (45 mg/lb) of body weight for severe infections. Since safety for use in premature infants and in neonates has not been established, the use of Cefazolin for Injection in these patients is not recommended. In pediatric patients with mild to moderate renal impairment (creatinine clearance of 70 to 40 mL/min), 60% of the normal daily dose given in equally divided doses every 12 hours should be sufficient. In patients with moderate impairment (creatinine clearance of 40 to 20 mL/min), 25% of the normal daily dose given in equally divided doses every 12 hours should be adequate. Pediatric patients with severe renal impairment (creatinine clearance of 20 to 5 mL/min) may be given 10% of the normal daily dose every 24 hours. All dosage recommendations apply after an initial loading dose. RECONSTITUTION Preparation of Parenteral Solution Parenteral drug products should be SHAKEN WELL when reconstituted, and inspected visually for particulate matter prior to administration. If particulate matter is evident in reconstituted fluids, the drug solutions should be discarded. Reconstituted solutions may range in color from pale yellow to yellow without a change in potency. Directions for Proper Use of a Pharmacy Bulk Package Not for direct infusion. This Pharmacy Bulk Package is for use in a hospital pharmacy admixture service only in a suitable work area, such as a laminar flow hood. Using aseptic technique, the container closure may be penetrated only one time after reconstitution using a suitable sterile dispensing set or transfer device that allows measured dispensing of the contents. Use of a syringe and needle is not recommended as it may cause leakage. The withdrawal of container contents should be accomplished without delay. However, should this not be possible, a maximum time of 4 HOURS from initial closure entry is permitted to complete fluid transfer operations. This time limit should begin with the introduction of the solvent or diluent into the Pharmacy Bulk Package. DISCARD ANY UNUSED PORTION AFTER 4 HOURS . Pharmacy Bulk Package Add Sterile Water for Injection, Bacteriostatic Water for Injection or Sodium Chloride Injection according to the table below. SHAKE WELL. Use promptly. (Discard vial within 4 hours after initial entry.) Pharmacy Bulk Package Size Amount of Diluent Appropriate Concentration Approximate Available Volume 10 grams 45 mL 1 gram/5 mL 51 mL 96 mL 1 gram/10 mL 102 mL 20 grams 87 mL 1 gram/5 mL 99 mL ADMINISTRATION Intravenous Administration Intermittent or continuous infusion: Dilute reconstituted Cefazolin for Injection, USP in 50 to 100 mL of one of the following solutions: Sodium Chloride Injection, USP 5% or 10% Dextrose Injection, USP 5% Dextrose in Lactated Ringer’s Injection, USP 5% Dextrose and 0.9% Sodium Chloride Injection, USP 5% Dextrose and 0.45% Sodium Chloride Injection, USP 5% Dextrose and 0.2% Sodium Chloride Injection, USP Lactated Ringer’s Injection, USP Invert Sugar 5% or 10% in Sterile Water for Injection Ringer’s Injection, USP 5% Sodium Bicarbonate Injection, USP When diluted according to the instructions above, cefazolin is stable for 24 hours at room temperature or for 10 days if stored under refrigeration (5ºC or 41ºF). Prior to administration parenteral drug products should be inspected visually for particulate matter and discoloration whenever solution and container permit.
