Drug Catalog - Product Detail
DORZOLAMIDE/ TIMOLOL OPHTHALMIC SOLUTION SOL 20/5MG/ML 10ML
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 50383-0233-10 | AKORN | 10 | 22.3-6.8MG/ML | SOLUTION |
PACKAGE FILES



Generic Name
Substance Name
Product Type
Route
Application Number
Description
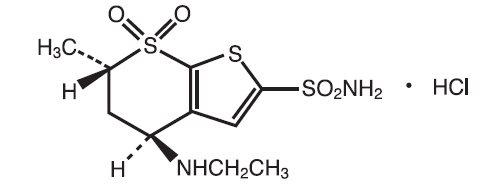
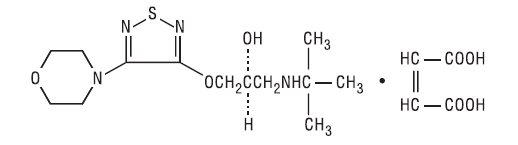
11 DESCRIPTION Dorzolamide Hydrochloride and Timolol Maleate Ophthalmic Solution is the combination of a topical carbonic anhydrase inhibitor and a topical beta-adrenergic receptor blocking agent. Dorzolamide hydrochloride is described chemically as: (4 S - trans )-4-(ethylamino)-5,6-dihydro-6-methyl-4 H -thieno[2,3- b ]thiopyran-2- sulfonamide 7,7-dioxide monohydrochloride. Dorzolamide hydrochloride is optically active. The specific rotation is: Its empirical formula is C 10 H 16 N 2 O 4 S 3 •HCl and its structural formula is: Dorzolamide hydrochloride has a molecular weight of 360.91. It is a white to off-white, crystalline powder, which is soluble in water and slightly soluble in methanol and ethanol. Timolol maleate is described chemically as: (-)-1-(tert-butylamino)-3-[(4- morpholino-1,2,5-thiadiazol-3-yl)oxy]-2-propanol maleate (1:1) (salt). Timolol maleate possesses an asymmetric carbon atom in its structure and is provided as the levo-isomer. The optical rotation of timolol maleate is: Its molecular formula is C 13 H 24 N 4 O 3 S•C 4 H 4 O 4 and its structural formula is: Timolol maleate has a molecular weight of 432.50. It is a white, odorless, crystalline powder which is soluble in water, methanol, and alcohol. Timolol maleate is stable at room temperature. Dorzolamide Hydrochloride and Timolol Maleate Ophthalmic Solution is supplied as a sterile, clear, colorless to nearly colorless, isotonic, buffered, slightly viscous, aqueous solution. The pH of the solution is approximately 5.65, and the osmolarity is 242-323 mOsM. Each mL of Dorzolamide Hydrochloride and Timolol Maleate Ophthalmic Solution contains 20 mg dorzolamide (22.26 mg of dorzolamide hydrochloride) and 5 mg timolol (6.83 mg timolol maleate). Inactive ingredients are sodium citrate, hydroxyethyl cellulose, sodium hydroxide, mannitol, and purified water. Benzalkonium chloride 0.0075% is added as a preservative. specific rotation chemical structure_dorzolamide optical rotation_timolol chemical structure_timolol
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING Dorzolamide Hydrochloride and Timolol Maleate Ophthalmic Solution is supplied sterile in 10 mL white LDPE plastic dropper bottles with white LDPE dropper tips and blue P/P caps of the following sizes: 5 mL 10 mL Storage Store Dorzolamide Hydrochloride and Timolol Maleate Ophthalmic Solution between 20 and 25°C (68-77°F). Protect from light. After opening, Dorzolamide Hydrochloride and Timolol Maleate Ophthalmic Solution can be used until the expiration date on the bottle.
Indications & Usage
1 INDICATIONS AND USAGE Dorzolamide hydrochloride and timolol maleate ophthalmic solution is indicated for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension who are insufficiently responsive to betablockers (failed to achieve target IOP determined after multiple measurements over time). The IOP-lowering of dorzolamide hydrochloride and timolol maleate ophthalmic solution administered twice a day was slightly less than that seen with the concomitant administration of 0.5% timolol administered twice a day and 2% dorzolamide administered three times a day [see Clinical Studies (14) ] . • Dorzolamide hydrochloride and timolol maleate ophthalmic solution is a combination of dorzolamide hydrochloride, carbonic anhydrase inhibitor, and timolol maleate, a beta-adrenergic receptor blocking agent, indicated for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension who are insufficiently responsive to beta-blockers. • The IOP-lowering of dorzolamide hydrochloride and timolol maleate ophthalmic solution twice daily was slightly less than that seen with the concomitant administration of 0.5% timolol twice daily, and 2% dorzolamide three times daily. (1)
Dosage and Administration
2 DOSAGE AND ADMINISTRATION The dose is one drop of dorzolamide hydrochloride and timolol maleate ophthalmic solution in the affected eye(s) two times daily. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five minutes apart [see Drug Interactions (7.3) ] . The dose is one drop of dorzolamide hydrochloride and timolol maleate ophthalmic solution in the affected eye(s) two times daily. (2)
