Drug Catalog - Product Detail
LAMIVUDINE OS 100MG/ML 240ML
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 57237-0274-24 | RISING PHARMACEUTICALS | 240 | 10MG/ML | SOLUTION |
PACKAGE FILES
Generic Name
LAMIVUDINE
Substance Name
LAMIVUDINE
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA077695
Description
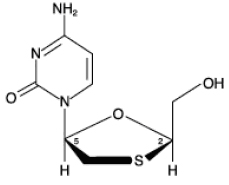
11 DESCRIPTION Lamivudine (also known as 3TC) is a synthetic nucleoside analogue with activity against HIV-1 and HBV. The chemical name of lamivudine is (2R,cis)-4-amino-l-(2-hydroxymethyl-l,3-oxathiolan-5-yl)-(1H)-pyrimidin-2-one. Lamivudine is the (-)enantiomer of a dideoxy analogue of cytidine. Lamivudine has also been referred to as (-)2',3'-dideoxy, 3'-thiacytidine. It has a molecular formula of C 8 H 11 N 3 O 3 S and a molecular weight of 229.3 g per mol. It has the following structural formula: Lamivudine USP is a white to off-white crystalline solid with a solubility of approximately 70 mg per mL in water at 20°C. Lamivudine oral solution USP is for oral administration. One milliliter (1 mL) of lamivudine oral solution USP contains 10 mg of lamivudine USP (10 mg per mL) in an aqueous solution and the inactive ingredients are liquid strawberry flavor, liquid banana flavor, citric acid (anhydrous), methylparaben, propylene glycol, propylparaben, sodium citrate (dihydrate), and sucrose (200 mg). Chemical Structure
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING Lamivudine Oral Solution USP is a clear, colorless to pale yellow, strawberry-banana flavored liquid, contains 10 mg of lamivudine in each 1 mL in HDPE bottles of 240 mL (NDC 57237-274-24) with child-resistant closures. This product does not require reconstitution. Recommended Storage: Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Indications & Usage
1 INDICATIONS AND USAGE Lamivudine oral solution is a nucleoside analogue indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus type 1 (HIV-1) infection. Limitations of Use: The dosage of this product is for HIV-1 and not for HBV. Lamivudine oral solution is a nucleoside analogue reverse transcriptase inhibitor indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection. Limitations of Use: The dosage of this product is for HIV-1 and not for HBV. (1)
Dosage and Administration
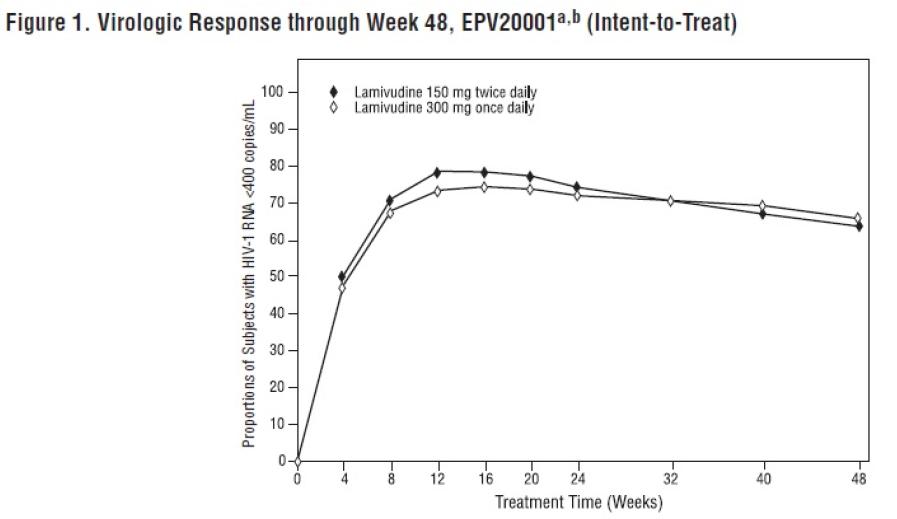
2 DOSAGE AND ADMINISTRATION Adults: 300 mg daily, administered as either 150 mg twice daily or 300 mg once daily. ( 2.1 ) Pediatric Patients Aged 3 Months and Older: Administered either once or twice daily. Dose should be calculated on body weight (kg) and should not exceed 300 mg daily. ( 2.2 ) Patients with Renal Impairment: Doses of lamivudine oral solution must be adjusted in accordance with renal function. ( 2.3 ) 2.1 Recommended Dosage for Adult Patients The recommended dosage of lamivudine oral solution in HIV-1-infected adults is 300 mg daily, administered as either 150 mg taken orally twice daily or 300 mg taken orally once daily with or without food. If lamivudine is administered to a patient infected with HIV-1 and HBV, the dosage indicated for HIV-1 therapy should be used as part of an appropriate combination regimen [see Warnings and Precautions (5.1) ]. 2.2 Recommended Dosage for Pediatric Patients Lamivudine scored tablet is the preferred formulation for HIV-1-infected pediatric patients who weigh at least 14 kg and for whom a solid dosage form is appropriate. Before prescribing lamivudine scored tablets, pediatric patients should be assessed for the ability to swallow tablets. For patients unable to safely and reliably swallow lamivudine tablets, the oral solution formulation may be prescribed [see Warnings and Precautions (5.5) ] . The recommended oral dosage of lamivudine tablets for HIV-1-infected pediatric patients is presented in Table 1. Table 1. Dosing Recommendations for Lamivudine Scored (150 mg) Tablets in Pediatric Patients Weight (kg) Twice-Daily Dosing Regimen Using Scored 150 mg Tablet Once-Daily Dosing Regimen a AM Dose PM Dose Total Daily Dose 14 to <20 1 tablet (150 mg) ½ tablet (75 mg) ½ tablet (75 mg) 150 mg ≥ 20 to <25 1½ tablets (225 mg) ½ tablet (75 mg) 1 tablet (150 mg) 225 mg ≥25 2 tablets (300 mg) b 1 tablet (150 mg) 1 tablet (150 mg) 300 mg a Data regarding the efficacy of once-daily dosing is limited to subjects who transitioned from twice-daily dosing to once-daily dosing after 36 weeks of treatment [see Clinical Studies (14.2)] . b Patients may alternatively take one 300 mg tablet, which is not scored. Oral Solution The recommended dosage of lamivudine oral solution in HIV-1-infected pediatric patients aged 3 months and older is 5 mg per kg taken orally twice daily or 10 mg per kg taken orally once daily (up to a maximum of 300 mg daily), administered in combination with other antiretroviral agents [see Clinical Pharmacology (12.3) ] . Consider HIV-1 viral load and CD4+ cell count/percentage when selecting the dosing interval for patients initiating treatment with oral solution [see Warnings and Precautions (5.5) , Clinical Pharmacology (12.3 )] . 2.3 Patients with Renal Impairment Dosing of lamivudine is adjusted in accordance with renal function. Dosage adjustments are listed in Table 2 [see Clinical Pharmacology (12.3) ] . Table 2. Adjustment of Dosage of Lamivudine in Adults and Adolescents (Greater than or Equal to 25 kg) in Accordance with Creatinine Clearance Creatinine Clearance (mL/min) Recommended Dosage of Lamivudine ≥50 150 mg twice daily or 300 mg once daily 30 to 49 150 mg once daily 15 to 29 150 mg first dose, then 100 mg once daily 5 to 14 150 mg first dose, then 50 mg once daily <5 50 mg first dose, then 25 mg once daily No additional dosing of lamivudine is required after routine (4-hour) hemodialysis or peritoneal dialysis. Although there are insufficient data to recommend a specific dose adjustment of lamivudine in pediatric patients with renal impairment, a reduction in the dose and/or an increase in the dosing interval should be considered.
