Drug Catalog - Product Detail
NITROFURANTOIN MONOHYDRATE/MACROCRYSTALS CAPS 100MG 100CT
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 16714-0439-01 | NORTHSTAR RX LLC | NA | NA | NA |
PACKAGE FILES

Generic Name
NITROFURANTOIN (MONOHYDRATE/MACROCRYSTALS)
Substance Name
NITROFURANTOIN
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA077066
Description
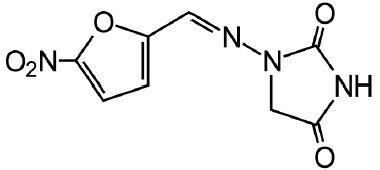
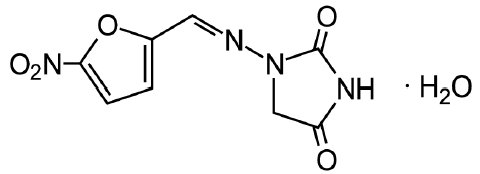
DESCRIPTION Nitrofurantoin is an antibacterial agent specific for urinary tract infections. Nitrofurantoin capsules, USP (monohydrate/macrocrystals) are hard gelatin capsule shells containing the equivalent of 100 mg of nitrofurantoin in the form of 25 mg of nitrofurantoin macrocrystals USP and 75 mg of nitrofurantoin monohydrate USP. The chemical name of nitrofurantoin macrocrystals USP is 1-[[[5-nitro-2-furanyl]methylene]amino]-2,4-imidazolidinedione. The chemical structure is the following: Molecular Weight: 238.16 The chemical name of nitrofurantoin monohydrate USP is 1-[[[5-nitro-2-furanyl]methylene]amino]-2,4-imidazolidinedione monohydrate. The chemical structure is the following: Molecular Weight: 256.17 Inactive Ingredients Each capsule contains carbomer 934P, colloidal silicon dioxide, corn starch, compressible sugar, D&C yellow No. 10, edible white ink, FD&C blue No. 1, FD&C red No. 40, gelatin, lactose monohydrate, magnesium stearate, povidone, talc, and titanium dioxide. Meets USP Dissolution Test 5 Chemical Structure 1 Chemical Structure 2
How Supplied
HOW SUPPLIED Nitrofurantoin capsules, USP (monohydrate/macrocrystals), for oral administration, are available as: 100 mg: Black and ivory opaque capsules imprinted “ E 122” on the cap and body and supplied as: NDC 16714-439-01 bottles of 100 NDC 16714-439-02 bottles of 1,000 Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Dispense contents in a tight, light-resistant container as defined in the USP with a child-resistant closure, as required. KEEP OUT OF THE REACH OF CHILDREN.
Indications & Usage
INDICATIONS AND USAGE Nitrofurantoin (monohydrate/macrocrystals) is indicated only for the treatment of acute uncomplicated urinary tract infections (acute cystitis) caused by susceptible strains of Escherichia coli or Staphylococcus saprophyticus. Nitrofurantoin is not indicated for the treatment of pyelonephritis or perinephric abscesses. To reduce the development of drug-resistant bacteria and maintain the effectiveness of nitrofurantoin (monohydrate/macrocrystals) and other antibacterial drugs, nitrofurantoin (monohydrate/macrocrystals) should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Nitrofurantoins lack the broader tissue distribution of other therapeutic agents approved for urinary tract infections. Consequently, many patients who are treated with nitrofurantoin (monohydrate/macrocrystals) are predisposed to persistence or reappearance of bacteriuria (see CLINICAL STUDIES ). Urine specimens for culture and susceptibility testing should be obtained before and after completion of therapy. If persistence or reappearance of bacteriuria occurs after treatment with nitrofurantoin (monohydrate/macrocrystals), other therapeutic agents with broader tissue distribution should be selected. In considering the use of nitrofurantoin (monohydrate/macrocrystals), lower eradication rates should be balanced against the increased potential for systemic toxicity and for the development of antimicrobial resistance when agents with broader tissue distribution are utilized.
Dosage and Administration
DOSAGE AND ADMINISTRATION Nitrofurantoin capsules (monohydrate/macrocrystals) should be taken with food. Adults and Pediatric Patients Over 12 Years One 100 mg capsule every 12 hours for seven days.
