Drug Catalog - Product Detail
NORETHINDRONE ACET/ ETHINYL ESTRADIOL BLISOVI FE 1MG/20MCG TB 3X28
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 68180-0865-13 | LUPIN PHARMACEUTICALS | 28 | 1-20MG-MCG | TABLET |
PACKAGE FILES



Generic Name
NORETHINDRONE ACETATE AND ETHINYL ESTRADIOL
Substance Name
Product Type
HUMAN PRESCRIPTION DRUG
Route
Application Number
ANDA201584
Description
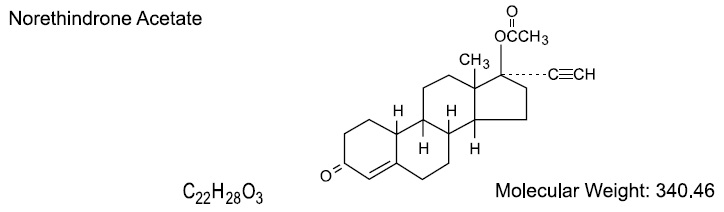
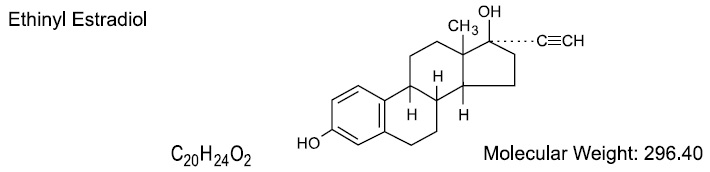
DESCRIPTION Blisovi™ Fe 1/20 is a progestogen-estrogen combination. Blisovi Fe 1/20 provides a continuous dosage regimen consisting of 21 oral contraceptive tablets and seven ferrous fumarate tablets. The ferrous fumarate tablets are present to facilitate ease of drug administration via a 28-day regimen, are nonhormonal, and do not serve any therapeutic purpose. Each yellow tablet contains norethindrone acetate (17α-ethinyl-19-nortestosterone acetate), 1 mg; ethinyl estradiol (17α-ethinyl-1,3,5(10)-estratriene-3,17β-diol), 20 mcg. Each yellow tablet contains the following inactive ingredients: acacia, confectioner's sugar, corn starch, D&C yellow no. 10 aluminium lake, lactose monohydrate, magnesium stearate, and talc. The structural formulas are as follows: Each brown placebo tablet contains ferrous fumarate, magnesium stearate, microcrystalline cellulose, povidone, sodium starch glycolate, and sucrose.The ferrous fumarate tablets do not serve any therapeutic purpose. *Ferrous fumarate tablets are not USP for dissolution. Norethindrone Acetate Ethinyl Estradiol
How Supplied
HOW SUPPLIED Blisovi Fe 1/20 ( norethindrone acetate and ethinyl estradiol tablets USP, 1 mg/0.02 mg and ferrous fumarate tablets) are available in a blister (NDC 68180-865-71) containing 28 tablets packed in a pouch (NDC 68180-865-71). Such three pouches are packaged in a carton (NDC 68180-865-73). Each blister contains 28 tablets, as follows: 21 yellow coloured, round flat face beveled edged tablets, each containing 1 mg norethindrone acetate and 0.02 mg ethinyl estradiol, debossed with "LU" on one side and "J23" on the other side. 7 brown mottled, round, flat face beveled edged tablets debossed with "LU" on one side and "M22" on the other side. Each brown tablet contains 75 mg ferrous fumarate. The ferrous fumarate tablets are present to facilitate ease of drug administration via a 28-day regimen, are non-hormonal, and do not serve any therapeutic purpose. Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature].
Indications & Usage
INDICATIONS AND USAGE Blisovi Fe 1/20 is indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception. Oral contraceptives are highly effective. Table I lists the typical accidental pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization, depends upon the reliability with which they are used. Correct and consistent use of methods can result in lower failure rates. Adapted from RA Hatcher et al, Reference 7. % of Women Experiencing an Unintended Pregnancy in the First Year of Continuous Use Method Lowest Expected The authors' best guess of the percentage of women expected to experience an accidental pregnancy among couples who initiate a method (not necessarily for the first time) and who use it consistently and correctly during the first year if they do not stop for any other reason. Typical This term represents "typical" couples who initiate use of a method (not necessarily for the first time), who experience an accidental pregnancy during the first year if they do not stop use for any other reason. (No contraception) (85) (85) Oral contraceptives 3 Combined 0.1 N/A N/A--Data not available Progestin only 0.5 N/A Diaphragm with spermicidal cream or jelly 6 20 Spermicides alone (foam, creams, gels, vaginal suppositories, and vaginal film) 6 26 Vaginal Sponge Nulliparous 9 20 Parous 20 40 Implant 0.05 0.05 Injection: depot medroxyprogesterone acetate 0.3 0.3 IUD Progesterone T 1.5 2.0 Copper T 380A 0.6 0.8 LNg 20 0.1 0.1 Condom without spermicides Female 5 21 Male 3 14 Cervical Cap with spermicidal cream of jelly Nulliparous 9 20 Parous 26 40 Periodic abstinence (all methods) 1 to 9 25 Withdrawal 4 19 Female sterilization 0.5 0.5 Male sterilization 0.10 0.15
Dosage and Administration
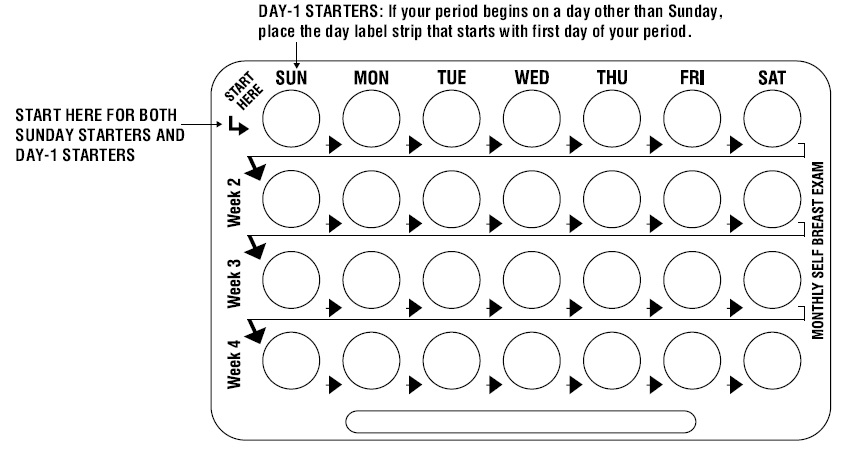
DOSAGE AND ADMINISTRATION The blister has been designed to make oral contraceptive dosing as easy and as convenient as possible. The tablets are arranged in four rows of seven tablets each, with the days of the week appearing on the blister above the first row of tablets. Note: Each blister has been preprinted with the days of the week, starting with Sunday, to facilitate a Sunday-Start regimen. Six different day label strips have been provided with the Detailed Patient & Brief Summary Patient Package Insert in order to accommodate a Day-1 Start regimen. If the patient is using the Day-1 Start regimen, she should place the self-adhesive day label strip that corresponds to her starting day over the preprinted days. Important: The patient should be instructed to use an additional method of protection until after the first week of administration in the initial cycle when utilizing the Sunday-Start regimen. The possibility of ovulation and conception prior to initiation of use should be considered. Dosage and Administration for 28-Day Dosage Regimen To achieve maximum contraceptive effectiveness, Blisovi Fe 1/20 should be taken exactly as directed and at intervals not exceeding 24 hours. Blisovi Fe 1/20 provides a continuous administration regimen consisting of 21 yellow tablets of norethindrone acetate and ethinyl estradiol, 1 mg/0.02 mg and 7 brown non-hormone containing tablets of ferrous fumarate. The ferrous fumarate tablets are present to facilitate ease of drug administration via a 28-day regimen and do not serve any therapeutic purpose. There is no need for the patient to count days between cycles because there are no "off-tablet days." 1. Sunday-Start Regimen: The patient begins taking the first yellow tablet from the top row of the blister (labeled Sunday) on the first Sunday after menstrual flow begins. When the menstrual flow begins on Sunday, the first yellow tablet is taken on the same day. The patient takes one yellow tablet daily for 21 days. The last yellow tablet in the blister will be taken on a Saturday. Upon completion of all 21 yellow tablets, and without interruption, the patient takes one brown tablet daily for 7 days. Upon completion of this first course of tablets, the patient begins a second course of 28-day tablets, without interruption, the next day (Sunday), starting with the Sunday yellow tablet in the top row. Adhering to this regimen of one yellow tablet daily for 21 days, followed without interruption by one brown tablet daily for seven days, the patient will start all subsequent cycles on a Sunday. 2 Day-1 Start Regimen: The first day of menstrual flow is Day 1. The patient places the self-adhesive day label strip that corresponds to her starting day over the preprinted days on the blister. She starts taking one yellow tablet daily, beginning with the first yellow tablet in the top row. After the last yellow tablet (at the end of the third row) has been taken, the patient will then take the brown tablets for a week (7 days). For all subsequent cycles, the patient begins a new 28 tablet regimen on the eighth day after taking her last yellow tablet, again starting with the first tablet in the top row after placing the appropriate day label strip over the preprinted days on the blister. Following this regimen of 21 yellow tablets and 7 brown tablets, the patient will start all subsequent cycles on the same day of the week as the first course. Tablets should be taken regularly with a meal or at bedtime. It should be stressed that efficacy of medication depends on strict adherence to the dosage schedule. Special Notes on Administration Menstruation usually begins two or three days, but may begin as late as the fourth or fifth day, after the brown tablets have been started. In any event, the next course of tablets should be started without interruption. If spotting occurs while the patient is taking yellow tablets, continue medication without interruption. If the patient forgets to take one or more yellow tablets, the following is suggested: One tablet is missed • take tablet as soon as remembered • take next tablet at the regular time Two consecutive tablets are missed (week 1 or week 2) • take two tablets as soon as remembered • take two tablets the next day • use another birth control method for seven days following the missed tablets Two consecutive tablets are missed (week 3) Sunday-Start Regimen : • take one tablet daily until Sunday • discard remaining tablets • start new pack of tablets immediately (Sunday) • use another birth control method for seven days following the missed tablets Day-1 Start Regimen : • discard remaining tablets • start new pack of tablets that same day • use another birth control method for seven days following the missed tablets Three (or more) consecutive tablets are missed Sunday-Start Regimen : • take one tablet daily until Sunday • discard remaining tablets • start new pack of tablets immediately (Sunday) • use another birth control method for seven days following the missed tablets Day-1 Start Regimen : • discard remaining tablets • start new pack of tablets that same day • use another birth control method for seven days following the missed tablets The possibility of ovulation occurring increases with each successive day that scheduled yellow tablets are missed. While there is little likelihood of ovulation occurring if only one yellow tablet is missed, the possibility of spotting or bleeding is increased. This is particularly likely to occur if two or more consecutive yellow tablets are missed. If the patient forgets to take any of the seven brown tablets in week four, those brown tablets that were missed are discarded and one brown tablet is taken each day until the pack is empty. A back-up birth control method is not required during this time. A new pack of tablets should be started no later than the eighth day after the last yellow tablet was taken. In the rare case of bleeding which resembles menstruation, the patient should be advised to discontinue medication and then begin taking tablets from a new blister on the next Sunday or the first day (Day-1), depending on her regimen. Persistent bleeding which is not controlled by this method indicates the need for reexamination of the patient, at which time nonfunctional causes should be considered. Use of Oral Contraceptives in the Event of a Missed Menstrual Period: 1. If the patient has not adhered to the prescribed dosage regimen, the possibility of pregnancy should be considered after the first missed period and oral contraceptives should be withheld until pregnancy has been ruled out. 2. If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out before continuing the contraceptive regimen. After several months on treatment, bleeding may be reduced to a point of virtual absence. This reduced flow may occur as a result of medication, in which event it is not indicative of pregnancy.
