Drug Catalog - Product Detail
RIZATRIPTAN BENZOATE TAB 10 MG (BASE EQUIVALENT) 18 CT
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 59746-0415-19 | JUBILANT CADISTA | 18 | 10MG | TABLET |
PACKAGE FILES



Generic Name
Substance Name
Product Type
Route
Application Number
Description
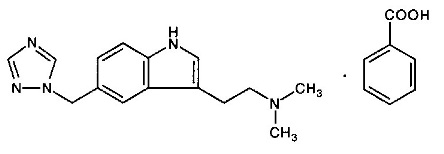
11 DESCRIPTION Rizatriptan benzoate tablets contains rizatriptan benzoate USP, a selective 5-hydroxytryptamine 1B/1D (5-HT 1B/1D ) receptor agonist. Rizatriptan benzoate USP is described chemically as: N,N -dimethyl-5-(1 H -1,2,4-triazol-1-ylmethyl)-1 H -indole-3-ethanamine monobenzoate and its structural formula is: Its molecular formula is C 15 H 19 N 5 •C 7 H 6 O 2 , representing a molecular weight of the free base of 269.4. Rizatriptan benzoate USP is a white to almost white, crystalline powder that is soluble in water and methanol, slightly soluble in isopropyl alcohol. Rizatriptan benzoate tablets are available for oral administration in strengths of 5 and 10 mg (corresponding to 7.265 mg or 14.53 mg of the benzoate salt, respectively). Each compressed tablet contains the following inactive ingredients: ferric oxide (red), lactose monohydrate, magnesium stearate, maize starch, and microcrystalline cellulose.
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING Rizatriptan benzoate tablets 5 mg are pale pink to pink colored, biconvex, capsule-shaped, uncoated tablets, debossed with ‘414’ on one side and ‘C’ on the other side. Unit-dose blister pack of 12 (2 x 6) tablets, NDC 59746-414-14 Unit-dose child-resistant blister pack of 18 (3 x 6) tablets, NDC 59746-414-19 Rizatriptan benzoate tablets 10 mg are pale pink to pink colored, biconvex, capsule-shaped, uncoated tablets, debossed with ‘415’ on one side and ‘C’ on the other side. Unit-dose blister pack of 12 (2 x 6) tablets, NDC 59746-415-14 Unit-dose child-resistant blister pack of 18 (3 x 6) tablets, NDC 59746-415-19 Storage Store rizatriptan benzoate tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
Indications & Usage
1 INDICATIONS AND USAGE Rizatriptan benzoate tablets are indicated for the acute treatment of migraine with or without aura in adults and in pediatric patients 6 to 17 years old. Limitations of Use Rizatriptan benzoate tablets should only be used where a clear diagnosis of migraine has been established. If a patient has no response for the first migraine attack treated with rizatriptan benzoate tablets, the diagnosis of migraine should be reconsidered before rizatriptan benzoate tablets are administered to treat any subsequent attacks. Rizatriptan benzoate tablets are not indicated for use in the management of hemiplegic or basilar migraine [see Contraindications (4)] . Rizatriptan benzoate tablets are not indicated for the prevention of migraine attacks. Safety and effectiveness of rizatriptan benzoate tablets have not been established for cluster headache. Rizatriptan benzoate is a serotonin (5-HT) 1B/1D receptor agonist (triptan) indicated for the acute treatment of migraine with or without aura in adults and in pediatric patients 6 to 17 years of age.( 1 ) Limitations of Use : Use only after clear diagnosis of migraine has been established ( 1 ) Not indicated for the prophylactic therapy of migraine ( 1 ) Not indicated for the treatment of cluster headache ( 1 )
Dosage and Administration
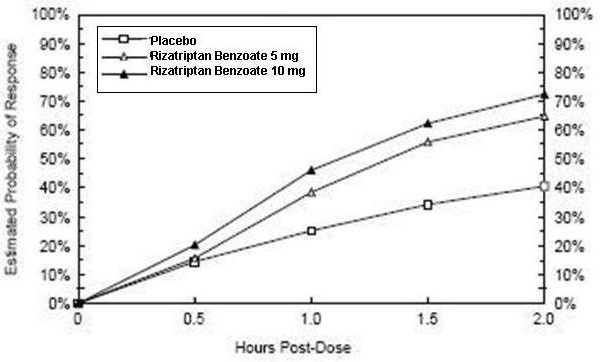
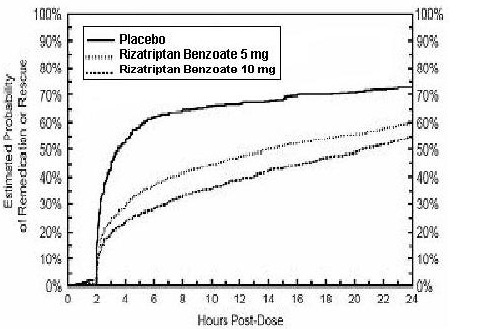
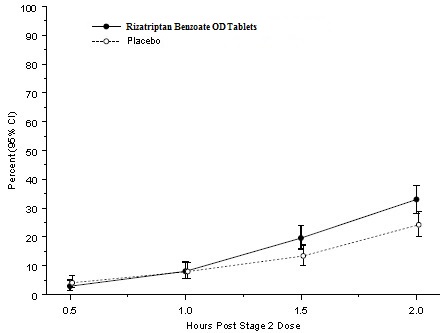
2 DOSAGE AND ADMINISTRATION Adults: 5 or 10 mg single dose; separate repeat doses by at least two hours; maximum dose in a 24-hour period: 30 mg (2.1) Pediatric patients 6 to 17 years: 5 mg single dose in patients less than 40 kg (88 lb); 10 mg single dose in patients 40 kg (88 lb) or more (2.2) Adjust dose if co-administered with propranolol ( 2.4) 2.1 Dosing Information in Adults The recommended starting dose of rizatriptan benzoate tablets is either 5 mg or 10 mg for the acute treatment of migraines in adults. The 10 mg dose may provide a greater effect than the 5 mg dose, but may have a greater risk of adverse reactions [see Clinical Studies (14.1) ] . Redosing in Adults Although the effectiveness of a second dose or subsequent doses has not been established in placebo-controlled trials, if the migraine headache returns, a second dose may be administered 2 hours after the first dose. The maximum daily dose should not exceed 30 mg in any 24-hour period. The safety of treating, on average, more than four headaches in a 30-day period has not been established. 2.2 Dosing Information in Pediatric Patients (Age 6 to 17 Years) Dosing in pediatric patients is based on the patient's body weight. The recommended dose of rizatriptan benzoate tablets is 5 mg in patients weighing less than 40 kg (88 lb), and 10 mg in patients weighing 40 kg (88 lb) or more. The efficacy and safety of treatment with more than one dose of rizatriptan benzoate tablets within 24 hours in pediatric patients 6 to 17 years of age have not been established. 2.4 Dosage Adjustment for Patients on Propranolol Adult Patients In adult patients taking propranolol, only the 5 mg dose of rizatriptan benzoate tablets is recommended, up to a maximum of 3 doses in any 24-hour period (15 mg) [see Drug Interactions (7.1) and Clinical Pharmacology (12.3) ]. Pediatric Patients For pediatric patients weighing 40 kg (88 lb) or more, taking propranolol, only a single 5 mg dose of rizatriptan benzoate tablets is recommended (maximum dose of 5 mg in a 24-hour period). Rizatriptan benzoate tablets should not be prescribed to propranolol-treated pediatric patients who weigh less than 40 kg (88 lb ) [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
