Drug Catalog - Product Detail
ZONISAMIDE CP 25MG 100
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 68462-0128-01 | GLENMARK PHARMACEUTICALS | 100 | 25MG | CAPSULE |
PACKAGE FILES





Generic Name
ZONISAMIDE
Substance Name
ZONISAMIDE
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA077651
Description
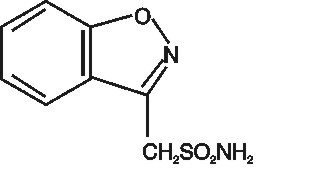
DESCRIPTION Zonisamide, USP is an antiseizure drug chemically classified as a sulfonamide and unrelated to other antiseizure agents. The active ingredient is zonisamide, USP, 1,2-benzisoxazole-3-methanesulfonamide. The empirical formula is C 8 H 8 N 2 O 3 S with a molecular weight of 212.23 g/mol. Zonisamide, USP is a white powder, pK a = 10.2, and is moderately soluble in water (0.80 mg/mL) and 0.1 N HCl (0.50 mg/mL). The chemical structure is: Zonisamide, USP is supplied for oral administration as capsules containing 25 mg, 50 mg or 100 mg zonisamide, USP. Each capsule contains the labeled amount of zonisamide, USP plus the following inactive ingredients: hydrogenated vegetable oil, microcrystalline cellulose, and sodium lauryl sulfate. The printed capsule shell of the different strengths is made from the following ingredients: 25 mg – D&C Red #28, FD&C Blue #1, gelatin and titanium dioxide 50 mg – D&C Yellow #10, FD&C Blue #1, FD&C Red #40, gelatin and titanium dioxide 100 mg – D&C Yellow #10, FD&C Blue #1, FD&C Yellow #6, gelatin and titanium dioxide The dyes used in the printing ink are FD&C Blue #1, FD&C Blue #2, FD&C Red #40, D&C Yellow #10 aluminum lake and iron oxide black. Additionally, the printing ink also contains propylene glycol and shellac. structure.jpg
How Supplied
HOW SUPPLIED Zonisamide capsules USP are available as 25 mg, 50 mg and 100 mg two-piece hard gelatin capsules. Zonisamide capsules USP are available in bottles of 100 and 500 with strengths and colors as follows: Dosage Strength Capsule Description Pack NDC # 25 mg White opaque body and light blue cap with ‘G 24’ printed on the cap and ‘25’ printed on the body in black ink. 100 count with child-resistant closure 68462-128-01 500 count 68462-128-05 50 mg White opaque body and yellow gold cap with ‘G 24’ printed on the cap and ‘50’ printed on the body in black ink. 100 count with child-resistant closure 68462-129-01 500 count 68462-129-05 100 mg White opaque body and light green cap with ‘G 24’ printed on the cap and ‘100’ printed on the body in black ink. 100 count with child-resistant closure 68462-130-01 500 count 68462-130-05 Store at 25°C (77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Store in a dry place and protect from light. Medication Guide available at www.glenmarkpharma-us.com/medguides Manufactured for: Glenmark Pharmaceuticals Inc., USA Mahwah, NJ 07430 Questions? 1 (888) 721-7115 www.glenmarkpharma-us.com October 2020 logo
Indications & Usage
INDICATIONS AND USAGE Zonisamide capsules are indicated as adjunctive therapy in the treatment of partial seizures in adults with epilepsy.
Dosage and Administration
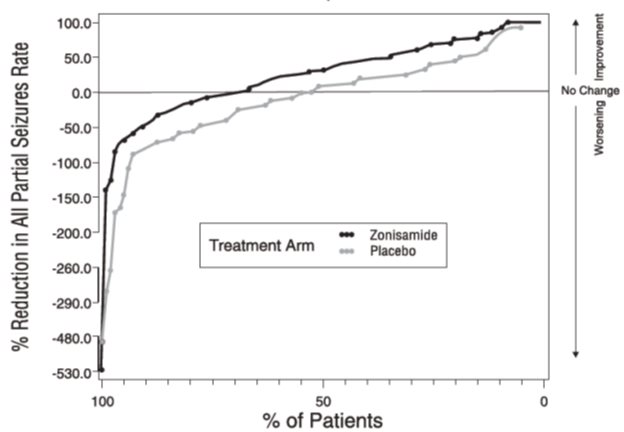
DOSAGE AND ADMINISTRATION Zonisamide capsules USP are recommended as adjunctive therapy for the treatment of partial seizures in adults. Safety and efficacy in pediatric patients below the age of 16 have not been established. Zonisamide capsules USP should be administered once or twice daily, using 25 mg or 100 mg capsules. Zonisamide capsules USP are given orally and can be taken with or without food. Capsules should be swallowed whole. Adults over Age 16: The prescriber should be aware that, because of the long half-life of zonisamide, up to two weeks may be required to achieve steady state levels upon reaching a stable dose or following dosage adjustment. Although the regimen described below is one that has been shown to be tolerated, the prescriber may wish to prolong the duration of treatment at the lower doses in order to fully assess the effects of zonisamide at steady state, noting that many of the side effects of zonisamide are more frequent at doses of 300 mg per day and above. Although there is some evidence of greater response at doses above 100 to 200 mg/day, the increase appears small and formal dose-response studies have not been conducted. The initial dose of zonisamide should be 100 mg daily. After two weeks, the dose may be increased to 200 mg/day for at least two weeks. It can be increased to 300 mg/day and 400 mg/day, with the dose stable for at least two weeks to achieve steady state at each level. Evidence from controlled trials suggests that zonisamide doses of 100 to 600 mg/day are effective, but there is no suggestion of increasing response above 400 mg/day (see CLINICAL PHARMACOLOGY, Clinical Studies subsection). There is little experience with doses greater than 600 mg/day. Patients with Renal or Hepatic Disease: Because zonisamide is metabolized in the liver and excreted by the kidneys, patients with renal or hepatic disease should be treated with caution, and might require slower titration and more frequent monitoring (see CLINICAL PHARMACOLOGY and PRECAUTIONS ).
